An international collaborative study with the participation of Prof. Cheng Teng‑Ip, assistant professor at the Centre for Cognitive and Brain Sciences (CCBS), University of Macau (UM), has applied deep learning algorithms to electroencephalogram (EEG) data from six independent global datasets worldwide. The team successfully distinguished patients with Major Depressive Disorder (MDD) from healthy controls and predicted treatment response to Selective Serotonin Reuptake Inhibitors (SSRIs) with nearly 80% accuracy. The findings have been published in Communications Medicine, a Nature Portfolio journal.
MDD ranks among the diseases with the highest global burden, yet current diagnostic methods remain largely subjective, and first‑line treatments exhibit a response rate of only about 50%. Patients often undergo prolonged trial‑and‑error medication cycles lasting weeks or months. While EEG is a low-cost, high-temporal-resolution, and widely available monitoring tool, it has long been underutilized in psychiatry. Although deep learning offers new opportunities for clinical EEG applications, previous studies were often limited to single datasets, making it difficult to validate model generalizability.
This study integrates six large, independent datasets from different laboratories around the world, encompassing eyes‑closed resting‑state EEG data from 146 healthy subjects and 203 MDD patients. The research team employed a Convolutional Neural Network (CNN) deep learning model using only 10‑channel EEG recordings (sampling rate 250 Hz) to distinguish patients from healthy controls and to predict SSRI treatment responders versus non‑responders. To ensure robustness, models were validated on completely unseen subject data. Additionally, Gradient-weighted Class Activation Mapping (Grad-CAM) was applied to identify the most discriminative neural features.
The model achieved an average accuracy of 67.5% (best fold 70%) in diagnosing MDD, with a sensitivity of 68.4% and specificity of 66.7%. For predicting SSRI response, it performed even better, achieving a mean accuracy of 79% (best fold: 85%), significantly outperforming 10 conventional machine learning algorithms and the EEGNet architecture. Grad-CAM analysis revealed that alpha-band activity (8–12 Hz) in frontal and parietal regions served as key biomarkers, with the right frontal F4 electrode carrying the highest weight. Clinical simulations showed that at 80% accuracy, the Number Needed to Treat (NNT) is five, potentially increasing the overall response rate from 50% to 70%.
These findings demonstrate the significant clinical potential of EEG‑based deep learning models, which may help psychiatrists identify patients most likely to benefit from SSRI treatment before treatment begins, thereby reducing ineffective trial periods and accelerating patient recovery. This approach requires only a low-cost 10-channel device, making it highly valuable for clinical application and providing new objective neurophysiological evidence for precision medicine and stratified treatment in psychiatry.
This research was supported by the University of Macau (SRG2023‑00040‑ICI & MYRG‑GRG2024‑00022‑ICI) and the Science and Technology Development Fund (FDCT) of Macau (0092/2025/ITP2).
The full version of the article can be viewed at: https://doi.org/10.1038/s43856-026-01394-z
Source: Centre for Cognitive and Brain Sciences, University of Macau
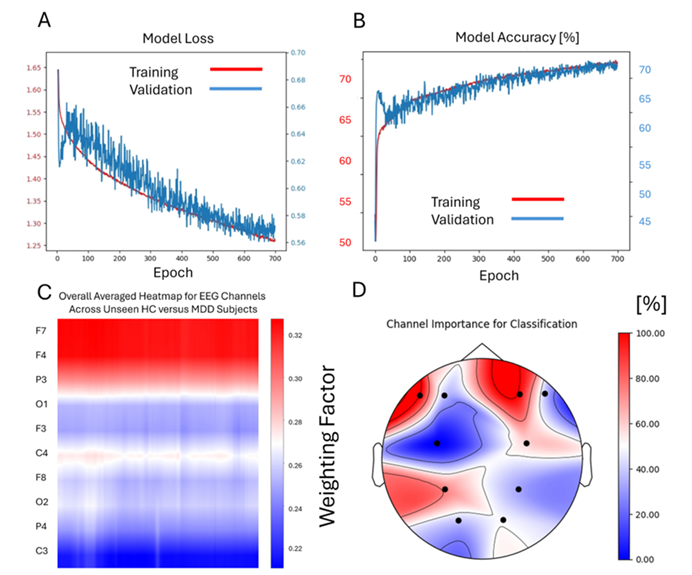
Figure 1
The training and validation process of the Deep Learning model for the discrimination of patients with MDD and Healthy Controls
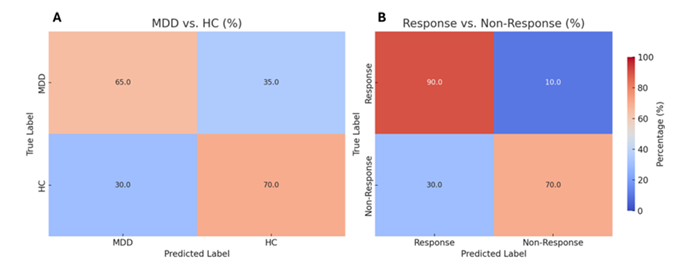
Figure 2
Confusion matrices for classification of patients with MDD versus Healthy Controls and for classification of Responders versus Non-Responders
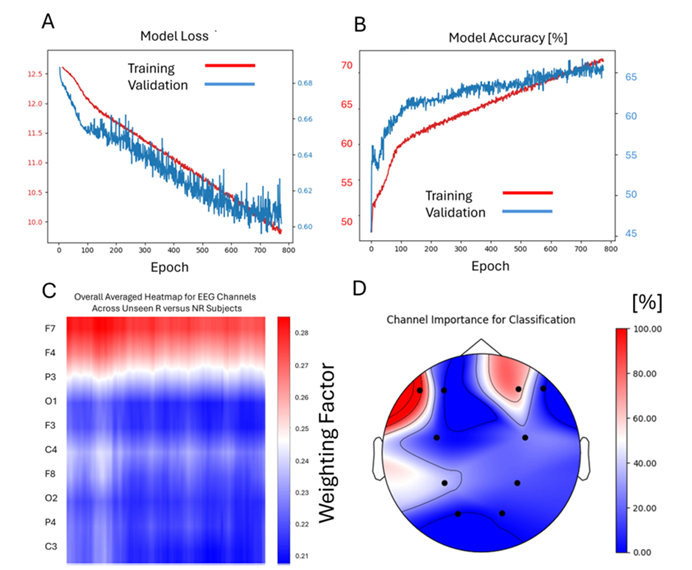
Figure 3
The training and validation process of the Deep Learning model for the discrimination of Responders and Non-Responders to SSRI treatment

